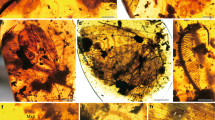
Abstract
Direct fossil evidence for Mesozoic flower pollination is scarce. Umenocoleoid micro-cockroaches Lepidopterix vegrandis gen. et sp. n. (Lebanese amber) and Antophiloblatta hispida gen. et sp. n. (Myanmar amber) possess size, form, complex coloration pattern and surface structure, cryptic with potentially entomophilous angiosperms Tropidogyne pentaptera Poinar, 2017 and Antiquifloris latifibris Poinar et Buckley, 2016 flower petals and sepals. Putative pollen grains attached to the latter adult indicates pollination, while reduced mouthparts suggest fluid nectar feeding. Spongistoma angusta gen. et sp. n. (Myanmar amber) has a narrow body and mandibles nearly entirely reduced with a unique “proboscis” forming sponging/ sucking mouthparts. In addition putative Classopolis Pflug, 1953 gymnosperm pollen is attached to adults and immature individuals of Vzrkadlenie miso gen. et sp. n. (Myanmar amber). Together with possible angiosperm pollination by Formicamendax vrsanskyi Hinkelman, 2019 and cycas pollination by immature individuals of alienopterid larvae, the evidence for early cockroach pollination is now substantial. Additionally unique is the forewing surface of L. vegrandis with photonic crystal structures within the scales.






Similar content being viewed by others
References
Azar D, Nel A, Geze R (2003) Use of Lebanese amber inclusions in paleoenvironmental reconstruction, dating and paleobiogeography. Acta Zool Cracov 46:393–398
Barnes WJP (2007) Functional morphology and design constraints of smooth adhesive pads. MRS Bull 32(6):479–485. https://doi.org/10.1557/mrs2007.81
Bryant D, Moulton V (2004) NeighborNet: an agglomerative algorithm for the construction of planar phylogenetic networks. Mol Biol Evol 21:255–265. https://doi.org/10.1007/3-540-45784-4_28
Chambers KL, Poinar G Jr, Buckley R (2010) Tropidogyne, a new genus of early cretaceous Eudicots (Angiospermae) from Burmese amber. Novon 20(1):23–29. https://doi.org/10.3417/2008039
Comstock JH, Needham JG (1898) The wings of insects. Am Nat 33:117–126
Cooke JAL (1968) A further record of predation by cockroaches Periplaneta americana. Entomol Mont Mag 104:1244–1246
Crepe WL, Nixon KC, Grimaldi D, Riccio M (2016) A mosaic Lauralean flower from the early cretaceous of Myanmar. Am J Bot 103(2):290–297. https://doi.org/10.3732/ajb.1500393
Dilcher DL, Basson PW (1990) Mid-cretaceous angiosperm leaves from a new fossil locality in Lebanon. Bot Gaz 151(4):538–547. https://doi.org/10.1086/337855
dos Santos Rolo T, Ershov A, van de Kamp T, Baumbach T (2014) In vivo X-ray cine-tomography for tracking morphological dynamics. Proc Natl Acad Sci USA 111:3291–3926. https://doi.org/10.1073/pnas.1308650111
Friis EM, Pedersen KR, Crane PR (2010) Diversity in obscurity: fossil flowers and the early history of angiosperms. Philos Trans R Soc Lond Ser B Biol Sci 365(1539):369–382. https://doi.org/10.1098/rstb.2009.0227
Friis EM, Crane PR, Pedersen KR (2011) Early flowers and angiosperm evolution. Cambridge University Press, Cambridge
Galant R, Skeath JB, Paddock S, Lewis DL, Carroll SB (1998) Expression pattern of a butterfly achaete-scute homolog reveals the homology of butterfly wing scales and insect sensory bristles. Curr Biol 8(14):807–813. https://doi.org/10.1016/S0960-9822(98)703227
Garrouste R, Hugel S, Jacquelin L, Rostan P, Steyer JS, Desutter-Grandcolas L, Nel A (2016) Insect mimicry of plants dates back to the Permian. Nat Commun 7:13735. https://doi.org/10.1038/ncomms13735
Ghiradella H, Aneshansley D, Eisner T, Silberglied RE, Hinton HE (1972) Ultraviolet reflection of a male butterfly: interference color caused by thin-layer elaboration of wing scales. Science 178(4066):1214–1217. https://doi.org/10.1126/science.178.4066.1214
Gorton RE Jr (1980) Comparative ecological study of the wood cockroaches in northeastern Kansas. Kansas Sci Bull 52:221–230
Grimaldi DA (1999) The co-radiations of pollinating insects and angiosperms in the cretaceous. Ann Missouri Bot 86:373–406. https://doi.org/10.2307/2666181
Grimaldi DA, Engel MS, Nascimbene PC (2002) Fossiliferous cretaceous amber from Myanmar (Burma): its rediscovery, biotic diversity, and paleontological significance. Am Mus Novit 3361:1–71. https://doi.org/10.1206/0003-0082(2002)361<0001:FCAFMB>2.0.CO;2
Hall R (2012) Late Jurassic–Cenozoic reconstructions of the Indonesian region and the Indian Ocean. Tectonophysics 570:1–41. https://doi.org/10.1016/j.tecto.2012.04.021
Hartkopf-Fröder C, Rust J, Wappler T, Friis EM, Viehofen A (2011) Mid-cretaceous charred fossil flowers reveal direct observation of arthropod feeding strategies. Biol Lett 8(2):295–298. https://doi.org/10.1098/rsbl.2011.0696
Hinkelman J (2020) Earliest behavioral mimicry and possible food begging in a Mesozoic alienopterid pollinator. Biologia 75:83–92. https://doi.org/10.2478/s11756-019-00278-z
Hu S, Dilcher DL, Jarzen DM, Taylor DW (2008) Early steps of angiosperm–pollinator coevolution. Proc Natl Acad Sci USA 105(1):240–245. https://doi.org/10.1073/pnas.0707989105
Huber BT, Norris RD, MacLeod KG (2002) Deep-sea paleotemperature record of extreme warmth during the cretaceous. Geology 30(2):123–126. https://doi.org/10.1130/0091-7613(2002)030<0123:DSPROE>2.0.CO;2
Ibrahim MI (2002) New angiosperm pollen from the upper Barremian-Aptian of the Western Desert, Egypt. Palynology 26(1):107–133. https://doi.org/10.2113/0260107
Jarzembowski EA (1994) Fossil cockroaches or pinnule insects? Proc Geologist Assoc 105(4):305–311. https://doi.org/10.1016/S0016-7878(08)80183-6
Khramov AV, Lukashevich ED (2019) A Jurassic dipteran pollinator with an extremely long proboscis. Gondwana Res 71:210–215. https://doi.org/10.1016/j.gr.2019.02.004
Kotulová J, Starek D, Havelcová M, Pálková H (2019) Amber and organic matter from the late Oligocene deep-water deposits of the Central Western Carpathians (Orava–Podhale Basin). Int J Coal Geol 207:96–109. https://doi.org/10.1016/j.coal.2019.02.006
Kraemer MMS, Kraemer AS, Stebner F, Bickel DJ, Rust J (2015) Entrapment bias of arthropods in Miocene amber revealed by trapping experiments in a tropical forest in Chiapas, Mexico. PLoS One 10(3):e0118820. https://doi.org/10.1371/journal.pone.0118820
Krassilov VA (1977) The origin of angiosperms. Bot Rev 43(1):143–176. https://doi.org/10.1007/BF02860852
Krassilov VA, Bacchia F (2000) Cenomanian florule of Nammoura, Lebanon. Cretac Res 21(6):785–799. https://doi.org/10.1006/cres.2000.0229
Krenn HW, Plant JD, Szucsich NU (2005) Mouthparts of flower-visiting insects. Arthropod Struct Dev 34(1):1–40. https://doi.org/10.1016/j.asd.2004.10.002
Kristensen NP, Simonsen TJ (2003) ‘Hairs’ and scales. Handbuch der Zoologie/ Handbook of Zoology 4(36):9–22
Labandeira CC (1998) How old is the flower and the fly? Science 280(5360):57–59. https://doi.org/10.1126/science.280.5360.57
Labandeira CC, Yang Q, Santiago-Blay JA, Hotton CL, Monteiro A, Wang YJ, Goreva Y, Shih C, Siljeström S, Rose T, Dilcher DL, Ren D (2016) The evolutionary convergence of mid-Mesozoic lacewings and Cenozoic butterflies. Proc R Soc B 283(1824):AN20152893. https://doi.org/10.1098/rspb.2015.2893
Lambert J, Johnson SC, Poinar GO (1996) Nuclear magnetic resonance characterization of cretaceous amber. Archaeometry 38(2):325–335. https://doi.org/10.1111/j.1475-4754.1996.tb00780.x
Latreille PA (1810) Considerations generales sur l'ordre naturel des animaux composant les classes des crustaces, des arachnides, et des insectes, avec un tableau methodique de leurs genres, disposes en familles. Chez. F. Schoell, Paris. https://doi.org/10.5962/bhl.title.39620
Li XR, Huang D (2018a) A new praying mantis from middle cretaceous Burmese amber exhibits bilateral asymmetry of forefemoral spination (Insecta: Dictyoptera). Cretac Res 91:269–273. https://doi.org/10.1016/j.cretres.2018.06.019
Li XR, Huang D (2018b) A new cretaceous cockroach with heterogeneous tarsi preserved in Burmese amber (Dictyoptera, Blattodea, Corydiidae). Cretac Res 92:12–17. https://doi.org/10.1016/j.cretres.2018.07.017
Liang J, Shih C, Wang L, Ren D (2019) New cockroaches (Insecta, Blattaria, Fuziidae) from the middle Jurassic Jiulongshan formation in northeastern China. Alcheringa 43:1–8. https://doi.org/10.1080/03115518.2019.1576061
Limaye A (2012) Drishti: a volume exploration and presentation tool. Proceedings SPIE 8506, developments in X-ray tomography VIII:85060X. https://doi.org/10.1117/12.935640
Luo ZX (2007) Transformation and diversification in early mammal evolution. Nature 450:1011–1019. https://doi.org/10.1038/nature06277
Maksoud S, Azar D, Granier B, Gèze R (2017) New data on the age of the lower cretaceous amber outcrops of Lebanon. Palaeoworld 26(2):331–338. https://doi.org/10.1016/j.palwor.2016.03.003
Metcalfe I (2013) Gondwana dispersion and Asian accretion: tectonic and palaeogeographic evolution of eastern Tethys. J Asian Earth Sci 66:1–33. https://doi.org/10.1016/j.jseaes.2012.12.020
Mlynský T, Wu H, Koubová I (2019) Dominant Burmite cockroach Jantaropterix ellenbergeri sp. n. might laid isolated eggs together. Palaeontogr Abt A 314:69–79. https://doi.org/10.1127/pala/2019/0091
Mullins DE (1984) Nutrient transfer in cockroaches. XVII International Congress of Entomology Abstract S(4)3.4:154
Nagamitsu T, Inoue T (1997) Cockroach pollination and breeding system of Uvaria elmeri (Annonaceae) in a lowland mixed-dipterocarp forest in Sarawak. Am J Bot 84(2):208–213. https://doi.org/10.2307/2446082
Negre B, Simpson P (2009) Evolution of the achaete-scute complex in insects: convergent duplication of proneural genes. Trends Genet 25(4):147–152. https://doi.org/10.1016/j.tig.2009.02.001
Nel A, Prokop J, Ross AJ (2008) New genus of leaf-mimicking katydids (Orthoptera: Tettigoniida), Orthoptera (Trias, NE France): mimétisme ou exaptation? PalZ 71(1–2):71–77. https://doi.org/10.1007/BF03022547
Peñalver E, Labandeira CC, Barrón E, Delclòs X, Nel P, Nel A, Tafforeau P, Soriano C (2012) Thrips pollination of Mesozoic gymnosperms. Proc Natl Acad Sci USA 109(22):8623–8628. https://doi.org/10.1073/pnas.1120499109
Peñalver E, Arillo A, Pérez-de la Fuente R, Riccio ML, Delclòs X, Barrón E, Grimaldi DA (2015) Long-proboscid flies as pollinators of cretaceous gymnosperms. Curr Biol 25(14):1917–1923. https://doi.org/10.1016/j.cub.2015.05.062
Peris D, Pérez-de la Fuente R, Peñalver E, Delclòs X, Barrón E, Labandeira CC (2017) False blister beetles and the expansion of gymnosperm-insect pollination modes before angiosperm dominance. Curr Biol 27(6):897–904. https://doi.org/10.1016/j.cub.2017.02.009
Podstrelená L, Sendi H (2018) Cratovitisma Bechly, 2007 (Blattaria: Umenocoleidae) recorded in Lebanese and Myanmar ambers. Paleontogr Abt A 310:121–129. https://doi.org/10.1127/pala/2018/0076
Poinar GO Jr (2004) Programinis burmitis gen. et sp. nov., and P. laminatus sp. nov., early cretaceous grass-like monocots in Burmese amber. Aust Syst Bot 17(5):497–504. https://doi.org/10.1071/SB04002
Poinar GO Jr (2017) A mid-cretaceous Lauraceae flower, Cascolaurus burmitis gen. et sp. nov., in Myanmar amber. Cretac Res 71:96–101. https://doi.org/10.1016/j.cretres.2016.11.015
Poinar GO Jr (2018) Burmese amber: evidence of Gondwanan origin and cretaceous dispersion. Hist Biol:1–6. https://doi.org/10.1080/08912963.2018.1446531
Poinar GO Jr, Milki R (2001) Lebanese amber. Oregon State University Press, Corvallis
Poinar Jr GO, Chambers KL (2005) Palaeoanthella huangii gen. and sp. nov., an early cretaceous flower (Angiospermae) in Burmese amber. SIDA Contributions to Botany 21:2087-2092
Poinar GO Jr, Chambers KL, Buckley R (2007a) Eoëpigynia burmensis gen. and sp. nov., an early cretaceous eudicot flower (Angiospermae) in Burmese amber. J Bot Res Inst Texas 1:91–96
Poinar GO Jr, Lambert JB, Wu Y (2007b) Araucarian source of fossiliferous Burmese amber: spectroscopic and anatomical evidence. J Bot Res Inst Texas 1(1):449–455
Poinar GO Jr, Chambers KL, Buckley R (2008) An early cretaceous angiosperm fossil of possible significance in rosid floral diversification. J Bot Res Inst Texas 2:1183–1192
Poinar GO Jr, Chambers KL, Wunderlich J (2013) Micropetasos, a new genus of angiosperms from mid-cretaceous Burmese amber. J Bot Res Inst Texas 7:745–750
Poinar GO Jr, Buckley R, Chen H (2016) A primitive mid-cretaceous angiosperm flower, Antiquifloris latifibris gen. & sp. nov., in Myanmar amber. J Bot Res Inst Texas 10:155–162
Poinar GO Jr, Chambers KL (2017) Tropidogyne pentaptera, sp. nov., a new mid-cretaceous fossil angiosperm flower in Burmese amber. Palaeodiversity 10(1):135–141. https://doi.org/10.18476/pale.v10.a10
Ren D, Labandeira CC, Santiago-Blay JA, Rasnitsyn A, Shih C, Bashkuev A, Logan MAV, Hotton L, Dilcher D (2009) A probable pollination mode befsore angiosperms: Eurasian, long-proboscid scorpionflies. Science 326(5954):840–847. https://doi.org/10.1126/science.1178338
Ross A, Mellish C, York P, Crighton B (2010) In: Penney D (ed) Biodiversity of fossils in Amber from the major world deposits. Siri Scientific Press, Manchester, pp 208–235
Santiago-Blay JA, Anderson SR, Buckley RT (2005) Possible implications of two new angiosperm flowers from Burmese amber (lower cretaceous) for well-established and diversified insect-plant associations. Entomol News 116(5):341
Saussure H (1864) Mémoires pour Servir a l’Histoire Naturelle du Mexique des Antilles et des États-Unis. Vol. 4. Orthoptères de l’Amérique Moyenne, Geneva, p 279
Schal C, Bell WJ (1982) Ecological correlates of paternal investment of urates in a tropical cockroach. Science 218(4568):170–173. https://doi.org/10.1126/science.218.4568.170
Schapheer C, Lopez-Uribe MM, Vera A, Villagra CA (2017) Distribution, habitat use and plant associations of Moluchia brevipennis (Saussure, 1864)(Blattodea: Ectobiidae): an endemic cockroach from Chilean Mediterranean Matorral biome. Rev Bras Entomol 61(2):114–122. https://doi.org/10.1016/j.rbe.2017.02.001
Scudder SH (1895) Revision of the American fossil cockroaches, with descriptions of new forms. Bulletin of the United States Geological and Geographical Survey of the Territories 124:1–176
Shi G, Grimaldi DA, Harlow GE, Wang J, Wang J, Yang M, Lei W, Li Q, Li X (2012) Age constraint on Burmese amber based on U–Pb dating of zircons. Cretac Res 37:155–163. https://doi.org/10.1016/j.cretres.2012.03.014
Sendi H, Azar D (2017) New aposematic and presumably repellent bark cockroach from Lebanese amber. Cretac Res 72:13–17. https://doi.org/10.1016/j.cretres.2016.11.013
Šmídová L, Lei X (2017) The earliest amber-recorded type cockroach family was aposematic (Blattaria: Blattidae). Cretac Res 72:189–199. https://doi.org/10.1016/j.cretres.2017.01.008
Swofford DL (2003) PAUP*. Phylogenetic analysis using parsimony (*and other methods) Ver. 4. Sinauer Associates, Sunderland
Sun G, Dilcher DL, Zheng S, Zhou Z (1998) In search of the first flower: a Jurassic angiosperm, Archaefructus, from northeast China. Science 282(5394):1692–1695. https://doi.org/10.1126/science.282.5394.1692
Uehara Y, Sugiura N (2017) Cockroach-mediated seed dispersal in Monotropastrum humile (Ericaceae): a new mutualistic mechanism. Bot J Linn Soc 185(1):113–118. https://doi.org/10.1093/botlinnean/box043
Vamosi JC, Vamosi SM (2010) Key innovations within a geographical context in flowering plants: towards resolving Darwin’s abominable mystery. Ecol Lett 13(10):1270–1279. https://doi.org/10.1111/j.1461-0248.2010.01521.x
Vidlička L, Vršanský P, Kúdelová T, Kúdela M, Deharveng L, Hain M (2017) New genus and species of cavernicolous cockroach (Blattaria, Nocticolidae) from Vietnam. Zootaxa 4232(3):361–375. https://doi.org/10.11646/zootaxa.4232.3.5
Vlasáková B, Kalinová B, Gustafsson MH, Teichert H (2008) Cockroaches as pollinators of Clusia aff. Sellowiana (Clusiaceae) on inselbergs in French Guiana. Ann Bot 102(3):295–304. https://doi.org/10.1093/aob/mcn092
Vogelgesang M, Chilingaryan S, dos Santos Rolo T, Kopmann A (2012) UFO: a scalable GPU-based image processing framework for on-line monitoring. Proceedings of the 14th IEEE International Conference on High Performance Computing and Communications, HPCC-2012 – 9th IEEE International Conference on Embedded Software and System ICESS-2012:824–829. https://doi.org/10.1109/HPCC.2012.116
Vršanský P (2003) Umenocoleoidea - an amazing lineage of aberrant insects (Insecta, Blattaria). AMBA Projekty 7:1–32
Vršanský P (2005) Mass mutations of insects at the Jurassic/Cretaceous boundary. Geol Carpath 56(6):473–781
Vršanský P, Liang JH, Ren D (2009) Advanced morphology and behaviour of extinct earwig-like cockroaches (Blattida: Fuziidae fam. nov.). Geol Carpath 60(6):449–462
Vršanský P, Oružinský R, Aristov D, Wei DD, Vidlička Ľ, Ren D (2017) Temporary deleterious mass mutations relate to originations of cockroach families. Biologia 72(8):886–912. https://doi.org/10.1515/biolog-2017-0096
Vršanský P, Bechly G, Zhang Q, Jarzembowski EA, Mlynský T, Šmídová L, Barna P, Kúdela M, Aristov D, Bigalk S, Krogmann L, Li L, Zhang Q, Zhang H, Ellenberger S, Müller P, Gröhn C, Xia F, Ueda K, Vďačný P, Valaška D, Vršanská L, Wang B (2018) Batesian insect-insect mimicry-related explosive radiation of ancient alienopterid cockroaches. Biologia 73(10):987–1006. https://doi.org/10.2478/s11756-018-0117-3
Vršanský P, Koubová I, Vršanská L, Hinkelman J, Kúdela M, Kúdelová T, Liang JH, Vidlička Ľ (2019a) Early wood-boring ‘mole roach’ reveals eusociality “missing ring”. AMBA Projekty 9(1):1–28
Vršanský P, Sendi H, Aristov D, Bechly G, Müller P, Ellenberger S, Azar D, Ueda K, Barna P, Garcia T (2019b) Ancient roaches further exemplify ‘no land return’in aquatic insects. Gondwana Res 68:22–33. https://doi.org/10.1016/j.gr.2018.10.020
Vršanský P, Šmídová L, Sendi H, Barna P, Müller P, Ellenberger S, Wu H, Ren X, Lei X, Azar D, Šurka J, Su T, Deng W, Shen X, Lv J, Bao T, Bechly G (2019c) Parasitic cockroaches indicate complex states of earliest proved ants. Biologia 74:65–89. https://doi.org/10.2478/s11756-018-0146-y
Vršanský P, Vršanská L, Beňo M, Bao T, Lei XJ, Ren XJ, Wu H, Šmídová L, Bechly G, Jun L, Yeo M, Jarzembowski E (2019d) Pathogenic DWV infection symptoms in a cretaceous cockroach. Paleontogr Abt A 314(1–3):1–10. https://doi.org/10.1127/0375-0442/2018/0000/0084
Vukusic P, Sambles JR (2003) Photonic structures in biology. Nature 424(6950):852. https://doi.org/10.1038/nature01941
Wang B, Xia F, Engel MS, Perrichot V, Shi G, Zhang H, Chen J, Jarzembowski EA, Wappler T, Rust J (2016) Debris-carrying camouflage among diverse lineages of Cretaceous insects. Sci Adv 2(6):e1501918. https://doi.org/10.1126/sciadv.1501918
Wedmann S, Bradler S, Rust J (2007) The first fossil leaf insect: 47 million years of specialized cryptic morphology and behavior. Proc Natl Acad Sci USA 104(2):565–569. https://doi.org/10.1073/pnas.0606937104
Wilhelmi AP, Krenn HW (2012) Elongated mouthparts of nectar-feeding Meloidae (Coleoptera). Zoomorphology 131(4):325–337
Winterton SL (2009) Scales and setae. In: Resh VH and Cardé RT (eds) Encyclopedia of Insects, 2nd edn. Academic Press, Cambridge, pp 901–904
Xu X, Zhou Z, Dudley R, Mackem S, Chuong CM, Erickson GM, Varricchio DJ (2014) An integrative approach to understanding bird. Science 346:2931. https://doi.org/10.1126/science.1253293
Acknowledgments
We thank reviewers for their valuable suggestions, Nikolaj Vršanský (private Bratislava), Bo Wang (NIGPAS Nanjing), Sieghard Ellenberger (private Germany), Tong Bao (University of Bonn) and Dany Azar (LU Bayeruth), Kamil Fekete (ESISAS Bratislava) for collecting and providing rare specimens and for technical help. We thank Elena Lukashevich (PINRAS Moscow) and Vladimir Blagoderov (NMS Edinburgh) for syninclusion determination. This work was supported by the Slovak Research and Development Agency under the contracts no. APVV-0436-12; VEGA 2/0139/17, 2/0042/18 and by UNESCO-Amba/ MVTS supporting grant of Presidium of the Slovak Academy of Sciences. Research at KIT was supported by the project UFO 2 (BMBF; 05K2012). We acknowledge the KIT light source for provision of instruments at their beamlines and we would like to thank the Institute for Beam Physics and Technology (IBPT) for the operation of the storage ring, the Karlsruhe Research Accelerator (KARA).
Author information
Authors and Affiliations
Contributions
P.V. (senior author) designed research and collected 2 adults in a local amber market; P.V., H.S. and J.H. collected data and wrote the paper; P.V., L.V. (V. miso including pseudo-inclusions, butterflies) H.S. (L. vegrandis, A. hispida) and J.H. (S. angusta) performed photo documentation, systematic evaluation and drawings of species; M.K. and T.K. performed phylogenetic analysis. M.Z. & T.v.d.K. performed synchrotron scans and post-processed tomographic data.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Article Zoobank
urn:lsid:zoobank.org:pub:674862A3-27D2-44EF-85C0-B31EF4A73C40
Rights and permissions
About this article
Cite this article
Sendi, H., Hinkelman, J., Vršanská, L. et al. Roach nectarivory, gymnosperm and earliest flower pollination evidence from Cretaceous ambers. Biologia 75, 1613–1630 (2020). https://doi.org/10.2478/s11756-019-00412-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-019-00412-x